Manual documentation stalls clinical innovation.
70%
Manual Document Processing
$1.8 B
Cost of Manual Work
42%
of all Deficiencies are Document-Related
12B
Lost Commercial Value
These inefficiencies stall the regulatory approvals of life-saving drugs and their delivery to patients
Scale CROs Revenue, Not Paperwork
70%
Efficiency Gain
25M
Hours of Manual Work Reclaimed
50 Days
Avoidable Delay
$540K
Daily Value Recovery
Al-Powered Platform Automating Regulatory Submissions
Sponsor Data Check
One-Click Generation
Compliance and multi-country localisation
Modifications Handling
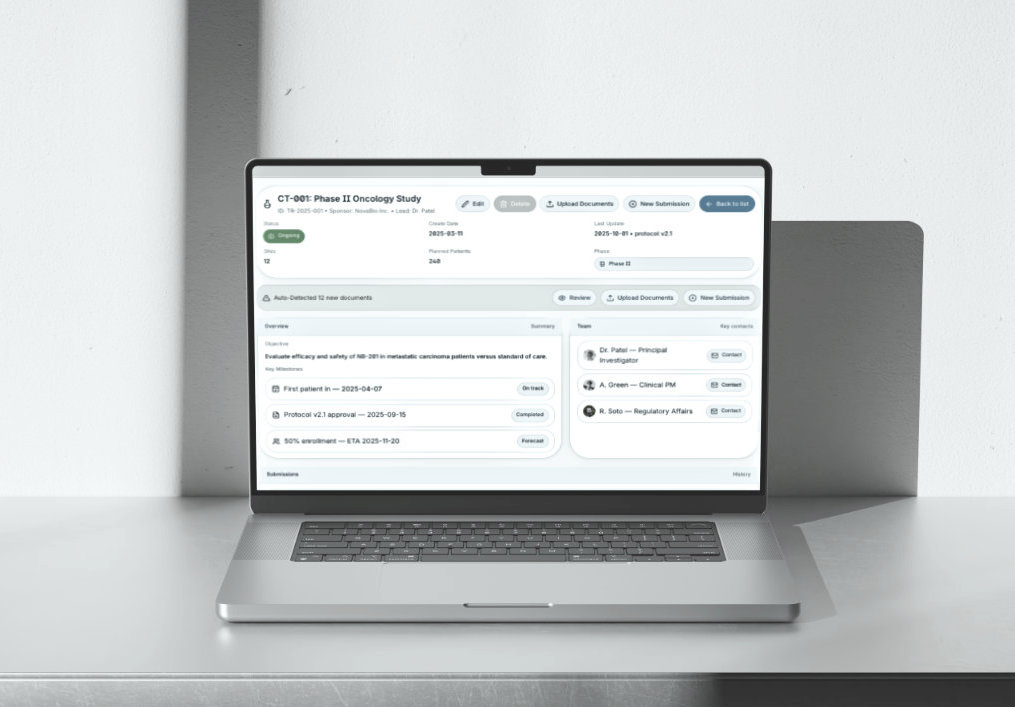
Trial Central Management Dashboard
Intelligent Search
powered by AI
Security & Human Control
Human-in-the-Loop
CRO regulatory teams are in control of every submission
Data Security and Privacy
Sovereign Infrastructure Built for Global Standards and sensitive data
Our Team of Experts

Pavel Sagulenko

Anna Hebenstreit
Corporate Lawyer with 15 + years of legal and compliance expereience in UK and Germany. Oversees legal compliance, operational excellence, and corporate governance to ensure seamless execution and compliance.

Ilya Billig
Validated by Enterprise Clients
Renewable Energy Company based in Munich
- Grant & Application Assistant | Produces accurate funding applications.
- Knowledge Base Agent | Enables smart search and instant data retrieval across all internal company data.
- Technical Writing Assistant | Generates compliant reports and manuals using templates.
Result: 70% reduction in writing cycles and the total elimination of manual „copy-paste“ errors.
Pharma Company based in Munich
- Protocol & Version Control | Tracking current and historical clinical trial protocols and modifications.
- Instant Search & History | Retrieval of company records from various applications.
Result: Accelerated clinical workflows through instant data access eliminating duplicate work.
Common Questions
Does your platform support clinical trials in any country?
Our platform is currently supporting submission in EU under CTIS and EU national legislation. It can be expanded to other countries on customer's demand.
What is your business and pricing model?
Our pricing is based on on the number of users and trials. Contact us to discuss your needs and pricing.
What makes your platform different from competition?
Unlike established e-clinical providers who offer generic clinical trial tools, our core focus is the generation of regulatory document packages compliant with CTIS and EU national laws.
Another key advantage of our platform is that it implements modifications across all relevant documents, consistently and error-free.
Unlike SaaS competitors who use public clouds, we offer a private German-hosted infrastructure or direct installation on your own servers. This ensures your sensitive trial data stays under your control.
Are you compliant with GDPR and EU AI Act?
The Platform is fully compliant with GDPR and EU AI Act.

